When veterinarians gave his rescue dog months to live, Paul Conyngham refused to accept the verdict. Armed with nothing but ChatGPT, Google’s AlphaFold, and relentless determination, the Australian tech entrepreneur did something unprecedented—he designed a custom mRNA cancer vaccine from scratch. The results stunned even the scientists who helped him build it.
The Beginning of an Impossible Quest
In the quiet suburbs of Sydney, Australia, a man sits in front of his computer at 2 AM, sifting through gigabytes of genetic data. His eyes are bloodshot. His coffee has gone cold hours ago. On the couch behind him, a seven-year-old staffy-Shar Pei cross named Rosie sleeps fitfully, her breathing occasionally labored, a tennis ball-sized tumor visible on her hind leg.
This scene repeated itself for months in 2024 and 2025. Paul Conyngham, a tech entrepreneur and data scientist with seventeen years of experience in machine learning, had been told by veterinarians that his beloved companion had only months to live. The cancer spreading through Rosie’s body—mast cell cancer, the most common skin cancer in dogs—had proven resistant to every conventional treatment. Surgery had been attempted multiple times. Chemotherapy had slowed the disease but couldn’t stop it. Immunotherapy had offered brief respite but ultimately failed.
But Paul Conyngham is not a man who accepts defeat easily. And what he did next would not only potentially save his dog’s life but would also send shockwaves through the medical and scientific community, raising profound questions about the future of personalized medicine, the democratization of biotechnology, and the role of artificial intelligence in healthcare.
He decided to build a cancer vaccine from scratch.
Without any formal training in biology.
Using AI tools available to anyone with an internet connection.
And against all odds, it worked.

The Man Behind the Mission
To understand how this seemingly impossible feat came to be, you first need to understand Paul Conyngham himself. He isn’t your typical tech entrepreneur chasing the next big exit. Based in Sydney, Conyngham has spent nearly two decades at the intersection of data science, artificial intelligence, and machine learning. He founded Core Intelligence, a company that develops AI solutions for clients ranging from startups to major banks, supermarket chains, insurance companies, and even the Australian Department of Defence.
He serves as a Director of the Data Science and AI Association of Australia (DSAI) and has been building GPT-derived systems for over four years—long before ChatGPT became a household name. He’s taught courses at General Assembly on AI-First Product Management and Data Analytics. He’s hosted workshops on algorithms like AlphaZero, delving into the intricacies of reinforcement learning, Monte Carlo tree search, and neural network design.
But for all his technical accomplishments, the most important thing in Paul’s life walked on four legs.
“Rosie is my best mate,” Paul told the Today Show in March 2026. “She’s been with me through really tough times, through a breakup, through hard business deals and walks in the forest. So when she was handed this sentence, I felt I had to do my part for her as well.”
Paul had adopted Rosie in 2019, a rescue dog who needed a home. Over the years, she became his constant companion, his confidante through the highs and lows of entrepreneurial life. When the cancer diagnosis came in 2024, it felt like the universe was delivering an impossibly cruel verdict.
But Paul had spent his career teaching machines to solve complex problems. If AI could help banks detect fraud, if it could help defense departments analyze threats, if it could help supermarket chains optimize their supply chains—could it also help him save his dog?

The Diagnosis That Changed Everything
Mast cell cancer is insidious. It’s the most common form of skin cancer in dogs, accounting for roughly 20% of all skin tumors. Mast cells are part of the immune system, normally helping the body respond to allergies and inflammation. But when they turn cancerous, they can be devastatingly aggressive.
The tumors appeared on Rosie’s back leg as lumps and nodules. Paul watched them grow despite thousands of dollars spent on veterinary chemotherapy. The treatment slowed the cancer’s spread, giving them precious time, but it couldn’t shrink the tumors. The veterinarians were running out of options.
“They told me she might have between one and six months left,” Paul recalled. “They said there wasn’t anything more they could do.”
For most pet owners, this would be the beginning of a painful goodbye, a period of making the most of limited time, of preparing for an inevitable loss. But Paul’s mind worked differently. He started asking questions.
What if the problem was that conventional treatments were too generic? What if the key to defeating Rosie’s cancer lay hidden within her own DNA? And what if the same AI tools he used every day for his clients could help him find answers that traditional veterinary medicine couldn’t provide?
Late one night, fueled by desperation and caffeine, Paul opened ChatGPT and typed his first query: How might I cure mast cell cancer in dogs?
Conversations with an AI
What followed was an unlikely collaboration between a determined dog owner and an artificial intelligence. Over weeks and months, Paul engaged in extensive conversations with ChatGPT, using the AI as a brainstorming partner, a research assistant, and a guide through unfamiliar scientific territory.
“ChatGPT assisted throughout that entire process,” Paul later explained. The AI helped him formulate a plan of attack, one that would require him to venture far outside his comfort zone as a data scientist and into the complex world of genomics and biotechnology.
The plan was audacious in its simplicity:
- Sequence Rosie’s DNA—both from healthy tissue and from her tumors
- Compare the two to identify the specific mutations causing the cancer
- Use AI tools to understand how those mutations affected the proteins in Rosie’s body
- Identify potential targets for treatment
- Design a personalized vaccine to attack the cancer
No one had ever done this for a dog before. The personalized mRNA cancer vaccines being developed by companies like Moderna and BioNTech were in clinical trials for humans, with billions of dollars invested and thousands of researchers working on the problems. Paul was one man with a laptop and a subscription to ChatGPT.
But he also had something those pharmaceutical companies didn’t: he had nothing to lose and everything to gain.
The $3,000 Gamble: Sequencing Rosie’s Genome
The first hurdle was practical. To identify the mutations driving Rosie’s cancer, Paul needed to sequence her DNA. This meant extracting genetic material from both her healthy blood cells and her tumor tissue, then processing that material through sophisticated sequencing machines that could read the billions of base pairs that made up her genome.
Paul reached out to the Ramaciotti Centre for Genomics at the University of New South Wales (UNSW), one of Australia’s premier genomic research facilities. The center had just celebrated its 25th anniversary and had contributed to significant projects like sequencing the bilby genome and analyzing skin cancers for the Australian Melanoma Genome Project.
Associate Professor Martin Smith, director of the center, was initially surprised by the request. It wasn’t every day that someone walked in asking to sequence their dog’s cancer genome. But Paul’s persistence—and his willingness to pay the $3,000 sequencing fee out of his own pocket—convinced Dr. Smith to help.
“He was a bit unusual,” Dr. Smith would later admit. But he was also impressed by Paul’s determination and his ability to articulate exactly what he needed.
During one of Rosie’s surgeries to remove tumors, a veterinarian collected tissue samples. These samples were sent to the Ramaciotti Centre, where the sequencing process began. The center’s state-of-the-art technology could process an entire human genome in just days for less than $1,000—a task that had cost billions of dollars and taken over a decade when the Human Genome Project first completed it in 2003.
Within weeks, Paul had approximately 350 gigabytes of raw genetic data sitting on his hard drive. It was, quite literally, Rosie written in code.
Now came the hard part: making sense of it all.

Reading the Code of Life with AI
Imagine receiving a book written in a language you don’t fully understand, containing three billion letters with no spaces between the words. Now imagine that somewhere in that book, hidden among all those letters, are a handful of typos that are causing your best friend’s body to destroy itself. Your job is to find those typos.
This is, in essence, what Paul Conyngham set out to do.
Using algorithms and data pipelines, Paul began comparing the DNA from Rosie’s healthy cells to the DNA from her tumor. He was looking for mutations—places where the genetic code in the cancer differed from the code in her healthy tissue. These mutations would be the fingerprints of the disease, the corrupted instructions that were telling Rosie’s cells to grow uncontrollably.
ChatGPT served as his guide through this process, helping him understand which tools to use, how to interpret the results, and where to focus his attention. For a man without a formal background in biology, it was like having a patient tutor available 24/7, one who never got tired of explaining the basics and never judged him for asking questions that a first-year genetics student might find elementary.
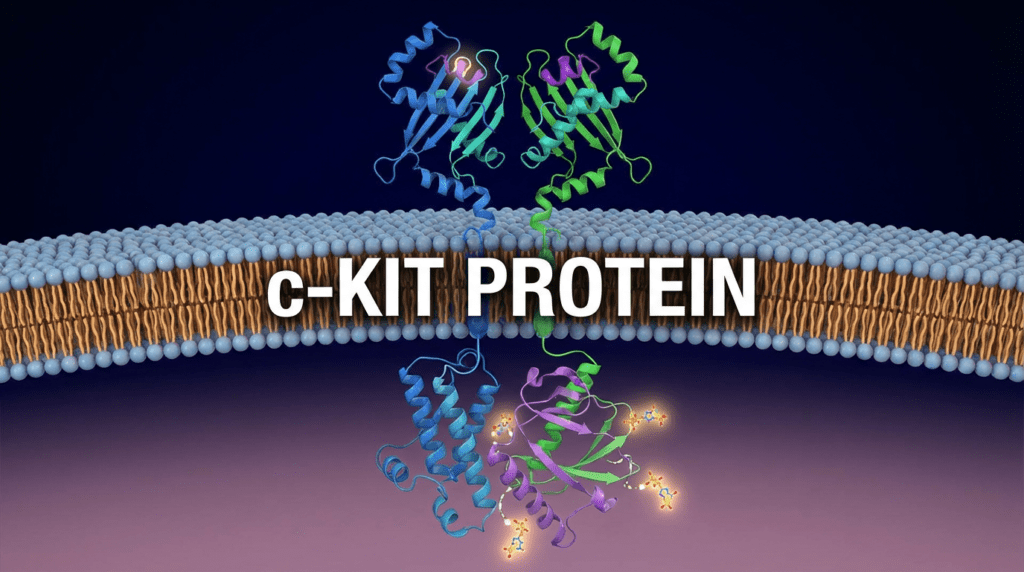
One mutation stood out: the c-KIT protein.
C-KIT is well-known in cancer research. It’s a receptor tyrosine kinase, a type of protein that sits on the surface of cells and helps regulate their growth and division. When the gene coding for c-KIT mutates, it can cause the protein to become permanently “switched on,” sending constant signals for cells to divide. This is a known driver of mast cell tumors in dogs.
Paul had found his target.
Enter AlphaFold: Seeing Proteins in Three Dimensions
Identifying the mutation was only half the battle. To design a treatment, Paul needed to understand how this mutated protein actually looked in three dimensions. Proteins aren’t flat blueprints; they’re complex molecular machines that fold into intricate shapes. The shape of a protein determines its function, and a mutation can change that shape in ways that make the protein behave abnormally.
Enter AlphaFold.
Developed by Google DeepMind, AlphaFold is an artificial intelligence system that represents one of the most significant scientific breakthroughs of the 21st century. The program can predict the three-dimensional structure of proteins from their amino acid sequences with remarkable accuracy—a problem that had stumped scientists for fifty years.
Before AlphaFold, determining a protein’s structure required painstaking experimental work using techniques like X-ray crystallography or cryo-electron microscopy. It could take months or years and cost hundreds of thousands of dollars to solve a single protein structure. AlphaFold could do it in hours.
In 2024, DeepMind’s Demis Hassabis and John Jumper received the Nobel Prize in Chemistry for AlphaFold, recognition of how profoundly the tool had transformed structural biology. The AlphaFold Protein Database now contains predicted structures for over 200 million proteins—essentially every protein known to science.
Paul fed his data into AlphaFold and watched as the AI rendered the three-dimensional structure of Rosie’s mutated c-KIT protein on his screen. For the first time, he could see exactly what was going wrong inside his dog’s body.
Dr. Kate Michie, a structural biologist at UNSW who has worked extensively with AlphaFold, later acknowledged the tool’s transformative impact while cautioning about its limitations. “AlphaFold has done amazing things,” she noted. “But it’s not perfect, and its predictions need to be validated experimentally.”
Still, for Paul’s purposes, AlphaFold provided something invaluable: insight. He could see the mutated protein. He could understand its vulnerabilities. And he could begin to design something to attack it.
Designing a Weapon: The mRNA Vaccine
The concept behind mRNA vaccines had become familiar to billions of people during the COVID-19 pandemic. The vaccines developed by Pfizer-BioNTech and Moderna used messenger RNA to instruct cells to produce a harmless piece of the coronavirus’s spike protein, teaching the immune system to recognize and attack the real virus if it ever appeared.
But the same technology could, in theory, be used against cancer.
Personalized cancer vaccines work on a similar principle. Instead of teaching the immune system to recognize a virus, they teach it to recognize the unique mutations present in a patient’s tumor. Since every cancer is genetically different—carrying its own constellation of mutations—the vaccine must be custom-designed for each patient.
Major pharmaceutical companies were already racing to develop this technology for humans. Moderna’s mRNA-4157, in combination with Merck’s immunotherapy drug pembrolizumab, was in Phase 3 clinical trials for melanoma and non-small cell lung cancer. BioNTech was running trials in pancreatic cancer, colorectal cancer, and head and neck cancers. Early results were promising—the melanoma trials showed a 44% reduction in the risk of recurrence or death.
But these trials had taken years to design. They had cost hundreds of millions of dollars. They required armies of scientists, regulatory experts, and clinical coordinators.
Paul Conyngham was one man with a laptop and a dying dog.
Using the data he’d gathered—Rosie’s mutations, the AlphaFold protein structures, his conversations with ChatGPT—Paul began designing a custom mRNA vaccine. The vaccine would encode pieces of Rosie’s mutated proteins, the neoantigens that marked her cancer cells as foreign. When injected, it would teach her immune system to recognize these markers and attack any cells displaying them.
It was, in essence, a precision-guided missile designed specifically for Rosie’s cancer.
Finding the Partners to Make It Real
Designing a vaccine on paper (or more accurately, in code) was one thing. Actually manufacturing it was another matter entirely.
Paul knew he needed help from experts who could translate his digital design into a physical medicine that could be injected into Rosie. He reached out to Páll Thordarson, director of the UNSW RNA Institute and a pioneer in nanomedicine.
Professor Thordarson was initially skeptical. Not of the science—he had worked with mRNA technology for years—but of the timeline.
“I was a bit skeptical at first, mainly due to concerns about the time it would take to develop such a vaccine,” he admitted later. In the pharmaceutical industry, developing a personalized cancer vaccine typically takes many months. Rosie might not have that long.
But when Professor Thordarson examined Paul’s data, he was impressed. The analysis was thorough. The target selection was sound. The vaccine design was viable.
“Once we had the sequence that Paul designed, it was less than two months until we handed it over to Paul, to the vet,” Professor Thordarson said. It was an astonishingly fast turnaround for what was, essentially, a first-in-class experimental treatment.
Associate Professor Martin Smith, the computational biologist who had initially helped sequence Rosie’s DNA, was “gobsmacked” by what Paul had accomplished. “His ability to analyze the complex genomic data himself with the aid of ChatGPT was remarkable,” Dr. Smith noted. This was someone with no formal training in biology who had managed to “crack the code” using publicly available AI tools.
The vaccine was manufactured in laboratories at UNSW. The RNA Institute used Paul’s specifications to synthesize the mRNA sequences, encapsulate them in lipid nanoparticles for delivery, and prepare doses for injection.
For the first time in history, a personalized cancer vaccine had been designed for a dog.

The Red Tape: Three Months of Bureaucracy
Ironically, the hardest part of the entire process wasn’t the science. It was the paperwork.
Australia, like most countries, has strict regulations governing experimental medical treatments—and for good reason. These regulations exist to protect patients (and animals) from unproven therapies that might cause more harm than good. But they weren’t designed for the scenario Paul found himself in: a desperate pet owner with a potentially life-saving treatment and a dog who didn’t have time to wait.
Obtaining ethical approval to administer an experimental vaccine to Rosie required Paul to prepare a comprehensive proposal—over 100 pages of documentation explaining the science behind the treatment, the potential risks and benefits, and the protocols for administering and monitoring the therapy.
This process took three months—longer than the vaccine itself had taken to design and manufacture.
Paul later reflected on the absurdity of the situation. He had used artificial intelligence to analyze a genome, predict protein structures, and design a personalized medicine in a matter of weeks. The bureaucracy required to actually use that medicine took longer than the scientific work itself.
Eventually, salvation came from an unexpected source. Mari Maeda, founder of the Canine Cancer Alliance in the United States, connected Paul with Professor Rachel Allavena, a canine immunotherapy researcher at the University of Queensland. Professor Allavena had existing ethical approvals for experimental immunotherapies in dogs, providing a pathway to finally treat Rosie.
Paul drove ten hours to deliver Rosie to the clinic where she would receive her first injection.
The Injection and the Wait
The first dose of the vaccine was administered in December 2025. A booster followed shortly after.
Then came the hardest part: waiting.
In early December, before the treatment began, Rosie’s condition had deteriorated significantly. The tumors on her leg had grown to the size of tennis balls. Her mobility was severely reduced. Paul described her as “shut down and a bit sad,” a shadow of the energetic companion who had bounded through forests alongside him for years.
Photos taken on August 27, 2025, showed Rosie’s leg covered in large tumors and skin lesions. By December 9, after just one week of treatment, the tumors were still evident, but there were signs of initial response. Paul watched anxiously, documenting every change, every subtle shift in Rosie’s behavior or physical condition.
Then something remarkable happened.
Within about a month of the first injection, one of Rosie’s tumors had shrunk by 75%. Another had halved in size. Her mobility began to return. Her energy levels increased. Her coat, which had grown dull during her illness, became glossy again.
By late January 2026—seven weeks into the trial—photos showed a dramatic improvement. The massive tumors that had disfigured Rosie’s leg had receded significantly. The skin had begun to heal.
“In early December, she was losing her mobility,” Paul recounted. “By the end of January, she was jumping over a fence to chase a rabbit.”
It wasn’t a complete cure. Some portions of the tumor didn’t respond to the treatment, suggesting the presence of additional mutations that the vaccine hadn’t targeted. Paul was already working on sequencing these resistant cells to understand why they had survived and to design a second vaccine to attack them.
But the improvement was undeniable. Rosie was alive. She was active. She was happy.
Against all odds, the experiment had worked.
The Scientific Reaction: “Citizen Science” Meets Cutting-Edge Medicine
When news of Paul’s achievement began to spread through the scientific community, the reaction was a mixture of astonishment, admiration, and cautious optimism.
David Thomas, inaugural director of the UNSW Centre for Molecular Oncology, highlighted the significance of what he called “citizen science.” Here was a non-scientist, without formal training in biology or medicine, who had used publicly available AI tools to accomplish something that typically required entire research teams and millions of dollars in funding.
“This demonstrates the potential for democratizing this technology,” Professor Thordarson observed. “We can actually do this here in Australia. We don’t have to necessarily rely on foreign companies to help us.”
Dr. Kate Michie, the structural biologist who had cautioned about AlphaFold’s limitations, nonetheless acknowledged the excitement of seeing non-scientists engage with these technologies. “This would have been unimaginable five years ago,” she noted.
But the scientists also issued important caveats. Cancer is extraordinarily complex. What worked for one dog with one type of cancer might not work for another. The partial response—with some tumors shrinking while others remained—illustrated the challenges that even personalized medicine faces against a disease that can evolve and adapt.
Professor Peter Bennett, a veterinary oncologist, emphasized the ongoing challenges in cancer treatment, including drug resistance and the need for rigorous validation of AI-predicted structures. The success with Rosie was remarkable, but it was also a single case study. Replicating and scaling this approach would require extensive additional research.
Still, the implications were clear. If one determined individual with access to publicly available AI tools could design an effective cancer vaccine for a dog, what might be possible when these technologies were applied more broadly?
Understanding the Science: How mRNA Vaccines Actually Work
To truly appreciate the elegance of what Paul accomplished, it’s worth understanding the science behind mRNA vaccines—a technology that went from obscure research subject to household term during the COVID-19 pandemic.
Every cell in your body contains DNA, the master blueprint of life. But DNA doesn’t directly make proteins; instead, it uses messenger RNA (mRNA) as an intermediary. Think of DNA as a precious original document stored in a vault (the cell nucleus), and mRNA as a photocopy that can be sent out to the factory floor (the ribosomes) where proteins are actually manufactured.
When you inject mRNA into the body, it enters cells and uses their machinery to produce whatever protein that mRNA encodes. In the case of COVID-19 vaccines, the mRNA encoded the spike protein of the coronavirus. Cells read the instructions, produced spike proteins, displayed them on their surfaces, and the immune system learned to recognize and attack anything bearing that spike.
Cancer vaccines work on the same principle, but with a crucial twist: instead of targeting a virus protein, they target proteins unique to cancer cells—specifically, the mutated proteins (neoantigens) that arise from the genetic errors driving the cancer.
Here’s why this is so powerful: cancer cells are, in essence, corrupted versions of your own cells. This makes them hard for the immune system to attack, because they look mostly “self.” But mutations create neoantigens—proteins that are genuinely foreign, markers that say “this cell is broken.” If you can teach the immune system to recognize these markers, you can potentially turn the body’s own defenses against the cancer.
The challenge has always been identification. Every patient’s cancer has its own unique set of mutations. There’s no one-size-fits-all neoantigen that works for everyone. This is why personalized cancer vaccines were, until recently, more theoretical than practical. The process of identifying each patient’s unique mutations, predicting which ones would make good vaccine targets, and manufacturing a custom vaccine was simply too slow and expensive.
AI changed this equation.
What Paul did was essentially replicate—in compressed form and with minimal resources—what pharmaceutical companies are spending billions to industrialize. He sequenced the tumor, identified the mutations, predicted the protein structures, selected the best targets, and designed the mRNA sequences that would encode them. Each step that once required teams of specialists could now be accelerated by artificial intelligence.
The vaccine he designed told Rosie’s cells to produce pieces of her cancer’s mutated proteins. Her immune system encountered these fragments, recognized them as foreign, and mounted a response. That response then extended to the cancer cells themselves, which bore the same mutated proteins.
It’s immunology meeting computer science meeting sheer determination—a convergence of fields that, a decade ago, would have seemed impossible to unite in someone’s home office.
The Emotional Weight: What Desperation Creates
There’s a deeper story here that the scientific details can obscure—the human story of desperation, love, and the lengths we go to for those we love.
Paul Conyngham didn’t set out to make medical history. He set out to save his friend.
Anyone who has watched a beloved pet deteriorate understands the particular anguish of helplessness. You can’t explain to a dog why they’re in pain, can’t comfort them with words about the future, can’t make promises you might not keep. All you can do is be present—and search, frantically, for anything that might help.
Paul described nights spent poring over research papers at 3 AM, trying to understand concepts that were entirely foreign to him. He described the emotional rollercoaster of hope and setback, of treatments that seemed to be working and then didn’t, of veterinarians who had done everything they could and had nothing left to offer.
“When she was handed this sentence, I felt I had to do my part for her as well,” he said simply.
That feeling—the refusal to accept defeat, the compulsion to do something even when the odds seem impossible—is a uniquely human trait. It’s what drives parents to advocate relentlessly for sick children, spouses to search for experimental treatments for dying partners, researchers to spend decades on problems everyone else has given up on.
But it’s also what can lead to tragedy. For every Paul Conyngham, there are countless others who have poured time and money into unproven treatments, who have been exploited by charlatans selling false hope, who have made desperate choices that brought only more suffering. The history of alternative medicine is littered with well-intentioned disasters.
What made Paul’s case different was the combination of genuine expertise (in data analysis, not biology, but expertise nonetheless), genuinely powerful tools (AI that could bridge knowledge gaps), and genuine scientific collaboration (the researchers at UNSW who validated and manufactured the vaccine). Hope was necessary, but it wasn’t sufficient. Hope had to be channeled through rigor.
The Collaboration That Made It Possible
Paul Conyngham’s story is often framed as a solo achievement—one man against cancer, armed with AI. But the reality is more nuanced and, in some ways, more inspiring.
He couldn’t have done it alone.
Associate Professor Martin Smith at the Ramaciotti Centre took a chance on an unusual request, sequencing a dog’s cancer genome when that wasn’t the center’s typical work. Professor Páll Thordarson at the UNSW RNA Institute provided the expertise to translate Paul’s data into an actual vaccine. Dr. Kate Michie offered guidance on interpreting AlphaFold’s predictions. Professor Rachel Allavena at the University of Queensland provided the ethical framework that allowed the treatment to proceed.
Mari Maeda, founder of the Canine Cancer Alliance in the United States, connected Paul with the resources he needed when Australian bureaucracy threatened to stall the project indefinitely. Veterinarians, lab technicians, and support staff all played roles in bringing the vaccine from concept to injection.
This collaborative dimension is crucial because it points toward how this kind of innovation might scale. Paul was an exceptional individual, but the system he navigated—universities, research institutes, regulatory bodies—was designed for a different era. Making personalized medicine accessible will require not just better AI but better infrastructure: streamlined approval processes, flexible manufacturing capabilities, networks of experts who can rapidly validate and implement novel treatments.
The scientists who helped Paul have spoken about how his case has changed their thinking. “What this taught me,” Professor Thordarson said, “is that we can actually do this here. We can democratize this technology in Australia.”
That democratization isn’t automatic. It requires intentional effort to make resources available, to lower barriers, to create pathways for motivated individuals and researchers to collaborate. But Paul’s case provides a template—an existence proof that the collaboration is possible and the results can be remarkable.
The Billion-Dollar Comparison: Big Pharma vs. One Man with a Chatbot
To fully appreciate what Paul Conyngham accomplished, it helps to understand what the pharmaceutical industry is doing in the same space—and how much it’s costing them.
Moderna, one of the world’s leading mRNA companies, is running Phase 3 clinical trials for its personalized cancer vaccine mRNA-4157 (also known as V940) in combination with Merck’s pembrolizumab. These trials involve hundreds of patients across multiple cancer types, including melanoma and non-small cell lung cancer. The development program has cost hundreds of millions of dollars and taken years to reach this point.
BioNTech, the German company that partnered with Pfizer on the COVID-19 vaccine, is running its own trials for personalized cancer vaccines in melanoma, pancreatic cancer, and colorectal cancer. The company has partnered with the UK’s National Health Service on an ambitious program aiming to provide personalized vaccines to up to 10,000 cancer patients by 2030.
Experts estimate that producing personalized cancer vaccines for humans costs between $100,000 and $300,000 per patient—a figure that reflects the complexity of rapid tumor sequencing, neoantigen prediction, mRNA synthesis, and quality control testing.
Paul Conyngham spent approximately $3,000 on sequencing, plus additional costs for vaccine manufacturing that he hasn’t publicly disclosed. While the total amount was “tens of thousands of dollars”—still significant—it’s a fraction of what large pharmaceutical companies are spending per patient.
The comparison isn’t entirely fair, of course. Pharmaceutical companies operate under stringent regulatory requirements, must prove safety and efficacy through rigorous clinical trials, and are building systems designed to scale to thousands or millions of patients. Paul was trying to save one dog.
But the contrast illustrates something important about the current moment in biotechnology. The tools that Paul used—DNA sequencing, ChatGPT, AlphaFold—were either free or relatively inexpensive. The knowledge he needed was available online. The bottleneck wasn’t access to technology; it was knowing how to use it.
This democratization of biotechnology represents a profound shift in how medical innovations might emerge. The next breakthrough might not come from a well-funded laboratory at a major pharmaceutical company. It might come from a garage, a bedroom, or a home office, created by someone with determination, curiosity, and access to AI.
The Future: From Dogs to Humans
The obvious question on everyone’s mind: if this worked for a dog, could it work for humans?
Professor Thordarson was unequivocal. “This world-first vaccine absolutely could be used to treat human cancer patients in the future,” he stated. The underlying technology—mRNA vaccines targeting cancer-specific mutations—is already being tested in human clinical trials. What Paul’s case demonstrated was that the process could be dramatically accelerated and simplified using AI tools.
Regulatory hurdles for human treatments are, of course, far more significant than those for veterinary medicine. Clinical trials require years of testing, extensive safety data, and approval from regulatory agencies like the FDA in the United States or the TGA in Australia. No amount of AI assistance can shortcut these requirements when human lives are at stake.
But the potential for AI to accelerate the discovery phase of this process is immense. Consider the steps Paul took:
- DNA sequencing: Already fast and relatively affordable
- Mutation identification: Accelerated dramatically by AI analysis
- Protein structure prediction: Transformed by AlphaFold
- Vaccine design: Guided by AI-assisted research and planning
Each of these steps used to take months or years. Now they can be completed in weeks. The rate-limiting factor is increasingly not the science itself, but the regulatory and manufacturing infrastructure needed to translate discoveries into treatments.
Companies are already working to address these bottlenecks. Moderna and BioNTech are developing manufacturing processes designed for rapid personalization. New AI tools are being created to optimize neoantigen selection and predict which mutations are most likely to generate an immune response. The infrastructure of personalized medicine is being built in real-time.
Experts now believe that personalized cancer vaccines could be available for some human patients as early as 2026 or 2027, with regulatory approvals potentially following within a year or two after that.
The Democratization of Biotechnology
Perhaps the most profound implication of Paul’s story isn’t medical at all—it’s social.
For most of human history, medical breakthroughs have been the domain of experts: trained physicians, laboratory scientists, pharmaceutical researchers. These people spent years in education and training, had access to specialized equipment and funding, and operated within institutions designed to support their work.
AI is changing this equation.
AlphaFold is freely available online. Anyone can use it. ChatGPT is accessible for a monthly subscription fee. DNA sequencing, while still requiring specialized laboratories, is commercially available and increasingly affordable. The knowledge required to understand genomics, protein biology, and vaccine design is documented in scientific papers that are increasingly available through open-access repositories.
This doesn’t mean that anyone can now design a cancer vaccine—the science is still complex, and Paul’s background in data analysis and machine learning gave him skills that most people don’t possess. But it does mean that the barriers to entry are lower than they’ve ever been.
Dr. Smith’s observation that Paul was able to “crack the code” without a biology background speaks to a fundamental shift in how expertise itself is being redefined. In the past, domain expertise was gatekept by years of formal education. Today, AI can serve as a bridge, helping intelligent, motivated individuals navigate unfamiliar territory.
This has implications far beyond medicine. What happens when motivated individuals can use AI to make breakthroughs in clean energy? In materials science? In agriculture? The same pattern of democratization that Paul demonstrated—combining publicly available AI tools with determination and domain-general problem-solving skills—could apply to countless fields.
The Risks and the Warnings
It would be irresponsible to tell this story without acknowledging the risks.
Not every attempt to treat cancer with homemade remedies ends well. For every Paul Conyngham, there may be others who lack the expertise to design safe treatments, who misinterpret AI outputs, or who pursue therapies that cause harm rather than healing. The regulations that delayed Rosie’s treatment exist for good reasons—they protect patients from the enthusiasm of well-meaning people who might inadvertently do damage.
The scientists involved in Paul’s project were careful to emphasize the caveats. Dr. Kate Michie warned that AlphaFold’s predictions need rigorous validation. Professor Bennett noted the challenges of drug resistance. The partial response to the vaccine—with some tumors responding while others didn’t—illustrated that even personalized medicine isn’t a silver bullet.
Cancer is not one disease; it’s hundreds of diseases, each with its own genetic profile and biological quirks. What worked for Rosie’s mast cell cancer might not work for another dog’s osteosarcoma or a human’s pancreatic cancer. Generalizing from a single success story is tempting but dangerous.
There’s also the question of access and equity. If personalized medicine requires tens of thousands of dollars, advanced AI tools, and the expertise to use them, it risks becoming available only to the wealthy and technically sophisticated. The democratization of biotechnology is meaningless if it only democratizes access for a privileged few.
These concerns are real and must be part of the conversation as these technologies mature. But they shouldn’t obscure the significance of what Paul achieved or the potential it represents.
Rosie Today
As of March 2026, Rosie is alive and thriving.
The tumors that once threatened to take her life have shrunk dramatically. Her energy has returned. She chases rabbits. She takes walks in the forest with Paul, just as she did before the diagnosis that seemed to seal her fate.
Paul continues to work on refining the treatment. He’s sequencing the portions of the tumor that didn’t respond to the first vaccine, trying to understand their resistance mechanisms. He’s collaborating with the scientists at UNSW to design a second vaccine that might address these remaining cancer cells.
He’s also thinking about how to help others in similar situations. The knowledge he gained through this process, the relationships he built with researchers, the protocols he developed—all of this could potentially benefit other pet owners facing similar diagnoses.
And beyond pets, there’s the larger vision. If personalized mRNA vaccines can be designed quickly and affordably using AI tools, and if the results with Rosie can be replicated in other cases, the implications for human medicine are staggering.
Professor Thordarson posed the question that many are now asking: “If we can do this for a dog, why aren’t we rolling this out to humans?”
The answer, for now, involves regulatory requirements, safety testing, and the need for rigorous clinical trials. But the gap between what’s technically possible and what’s clinically available is narrowing every day.
A Glimpse of the Future
Imagine a world where a cancer diagnosis doesn’t trigger despair but initiates a rapid, personalized response. Within days, your tumor is biopsied and sequenced. AI systems analyze your unique mutations, predict protein structures, and design a custom vaccine targeted specifically at your cancer. The vaccine is manufactured within weeks and administered alongside other treatments.
This isn’t science fiction. The pieces of this future already exist. DNA sequencing is fast and cheap. AI can analyze genomic data and predict protein structures. mRNA vaccine technology has been proven on a global scale. The question is no longer whether this future is possible but how quickly it will arrive and how widely it will be accessible.
Paul Conyngham and Rosie represent a proof of concept—not a finished product, but a glimpse of what’s coming. One determined individual, armed with AI tools and a refusal to accept the unacceptable, achieved something that would have seemed impossible just a few years ago.
The pharmaceutical industry is investing billions of dollars to build the infrastructure for personalized cancer vaccines at scale. The regulatory frameworks are evolving to accommodate these new therapies. The AI tools are improving with each passing month.
And somewhere in Sydney, a rescue dog who was supposed to be dead by now is chasing rabbits in the sunshine.
What This Means for Pet Owners and Patients
For the millions of pet owners who face similar diagnoses—that terrible moment when a veterinarian says “there’s nothing more we can do”—Paul’s story offers both hope and a cautionary lesson.
The hope is real. The tools Paul used are not locked away in classified laboratories. AlphaFold is free and publicly accessible. ChatGPT is available to anyone with a subscription. DNA sequencing services exist that will sequence any sample for a fee. The knowledge is out there, documented in scientific papers, tutorials, and forums.
But the cautionary lesson is equally important. Paul succeeded not just because he had access to these tools, but because he had the skills to use them correctly, the resources to fund the sequencing and manufacturing, the persistence to navigate months of bureaucracy, and—crucially—the collaboration of actual scientists who could validate his work and produce a vaccine that was safe to inject.
Attempting to replicate Paul’s success without this combination of factors could be dangerous. Misinterpreted data, poorly designed vaccines, or treatments administered outside of appropriate medical oversight could cause serious harm. The same AI tools that can accelerate discovery can also lead non-experts astray if used without appropriate validation.
For human patients, the lessons are similar. Personalized cancer vaccines are coming—probably sooner than most people realize. The clinical trials being run by Moderna, BioNTech, and others suggest that within a few years, some patients with certain cancers may have access to treatments designed specifically for their tumors.
But the path from a promising case study (whether canine or human) to widely available treatment is long and complex. Regulatory approval requires rigorous proof of safety and efficacy across many patients. Manufacturing must be scaled to meet demand. Healthcare systems must adapt to accommodate truly personalized treatments that can’t be mass-produced in advance.
The best thing patients and pet owners can do right now is stay informed, maintain relationships with qualified medical professionals, and advocate for the research and infrastructure that will make these treatments accessible. Paul’s story shows what’s possible; making it routine will require systemic change.
Conclusion: The Audacity of Hope
There’s a tendency in our culture to be cynical about technology, to focus on the downsides of AI, to worry about jobs lost and privacy invaded and misinformation spread. Those concerns are valid and important.
But Paul Conyngham’s story reminds us of something equally true: technology can be a force for profound good. The same AI systems that critics warn about were used by one man to save the life of his best friend. The same data analysis tools that drive corporate profits were repurposed to design a cancer vaccine. The same internet that spreads conspiracy theories also provides access to the accumulated knowledge of humanity.
What Paul did required courage—the courage to attempt something unprecedented, to enter a field he didn’t fully understand, to risk failure and heartbreak in pursuit of a chance at success. It required persistence—months of late nights analyzing data, navigating bureaucracy, and refusing to give up when obstacles appeared. And it required love—the kind of love that makes you look at a dying friend and say, “Not yet. Not if I can help it.”
The pharmaceutical executives running billion-dollar clinical trials have their own motivations, their own pressures, their own constraints. The regulatory officials reviewing experimental treatments have their own responsibilities. The scientists advancing personalized medicine have their own methods.
But sometimes progress comes from unexpected directions. Sometimes it comes from a tech entrepreneur who refuses to let his dog die. Sometimes one person with a chatbot can show us what’s possible before the institutions catch up.
Rosie’s story isn’t over. Neither is the story of personalized medicine, or of AI in healthcare, or of the ongoing effort to defeat cancer. But this chapter—the chapter of one man, one dog, and one audacious attempt to use AI against an incurable disease—has ended more happily than anyone dared to hope.
And it’s just the beginning.
A Note on Verification
The details in this story have been verified through multiple sources, including interviews conducted by the Australian Broadcasting Corporation, the Today Show Australia, The Australian newspaper, and official communications from the University of New South Wales. Professor Páll Thordarson, Associate Professor Martin Smith, and other researchers involved in the project have confirmed the key facts of the case.
While Rosie’s improvement has been dramatic and medically documented, it’s important to note that this remains a single case study. The scientific community awaits further research and replication before drawing broader conclusions about the efficacy of this approach.
Paul Conyngham continues to work with researchers at UNSW and other institutions on personalized cancer treatments. Rosie continues to chase rabbits. The future of medicine continues to be written—one patient, one mutation, one vaccine at a time.